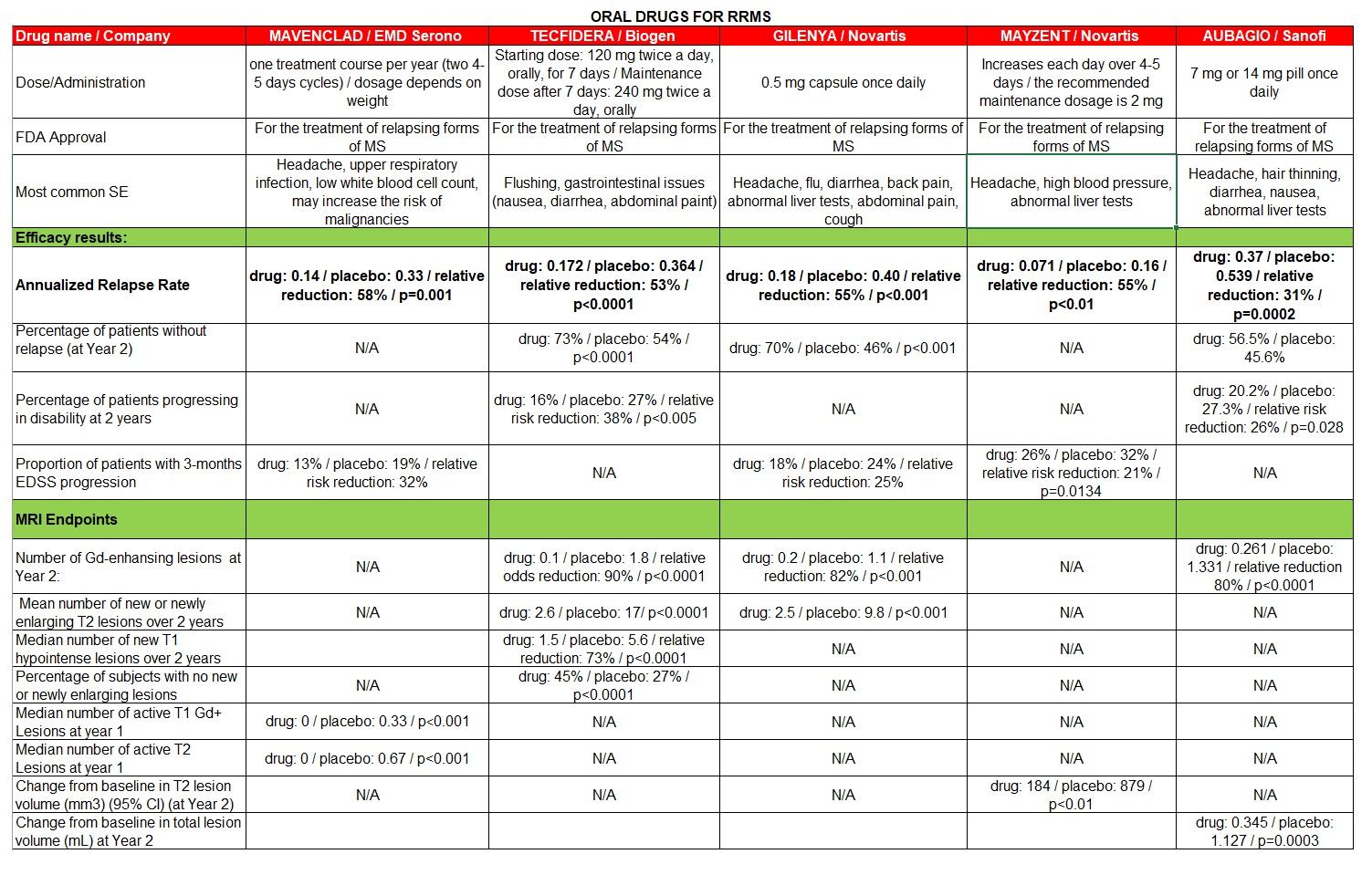
To develop the South San Francisco-based biotechs oral multiple sclerosis drug PRN2246. Sanofi also announced that it is planning to conduct four Phase 3 trials in both relapsing and progressive MS patients.
November 22 2021 Last Verified.

Btki sanofi ms. Sanofi also showcased data for its experimental MS drug tolebrutinib an oral brain-penetrant Brutons tyrosine kinase BTK inhibitor. Sanofis BTK inhibitor will potentially be first disease-modifying therapy to address sources of multiple sclerosis MS damage in the brain Sanofi to initiate four Phase 3 clinical trials in relapsing and progressive forms of MS. One of Sanofis top mid-stage drug prospects the subject of a recent short attack is being flagged through to a battery of pivotal Phase III.
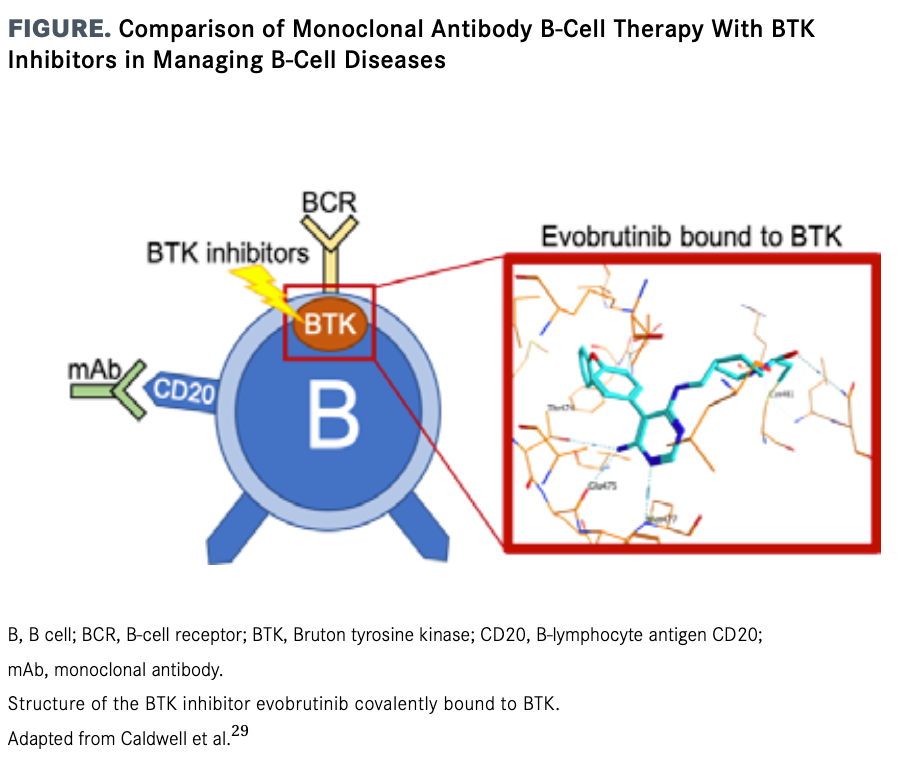
Sanofi scientists are investigating ways to target an enzyme called Brutons tyrosine kinase BTK. Novartis however notched a big win with another type of B-cell treatment in August when the FDA approved its Kesimpta. Brutons tyrosine kinase BTK inhibition is a novel therapeutic approach for treatment of multiple sclerosis MS given BTKs role in intracellular signaling of B lymphocytes and myeloid cells.
B-cell antigen receptor signaling. It stands as the only. BTK is critical to the activation of many immune cells that drive MS such as B cells and microglia and has gained attention for the role it plays in immune activities on both sides of the bloodbrain barrier.
Data from a Phase IIb long-term extension study in RMS showed that following 48 weeks of treatment tolebrutinib reduced multiple sclerosis disease activity as measured by MRI. December 9 2021 Last Verified. Sanofis BTK inhibitor flunks phase 3 autoimmune trial delivering blow to 37B biotech bet Sanofi still believes the rilzabrutinib clinical.
Plan to Share IPD. NCT04410991 Other Study ID Numbers. The takeover will give Sanofi full ownership of a BTK inhibitor it moved into a raft of phase 3 multiple sclerosis trials earlier this year.
Median number of prior therapies was 4. Global Medical Director- BTKi at Sanofi Genzyme San Antonio Texas United States 500 connections. MS Neuro Vaccines Cardiovascular metabolism R Registrational Study other than Phase 3 O Opt-in rights products for which rights have not been exercised yet O R O 1 Developed in collaboration with Immunext 2 Regeneron product for which Sanofi has opt-in rights 3 Developed in collaboration with Revolution Medicines 4 Developedin collaboration with.
Other than as required by applicable law Sanofi does not undertake any obligation to update or revise any forward-looking information or statements. Pre-BTKi cohort characteristics are summarized in Table 1. In April Sanofi presented phase 2.
Sanofi intends to initiate two Phase 3 trials in relapsing MS as well as two for rarer forms of the disease known as primary and secondary progressive in the middle of this year. Sanofi brain-penetrant BTK inhibitor meets primary endpoint of Phase 2 trial in relapsing multiple sclerosis Sanofis BTK inhibitor will potentially be first disease-modifying therapy to address sources of multiple sclerosis MS damage in the brain Sanofi to initiate four Phase 3 clinical trials in relapsing and progressive forms of MS. UTN First Posted.
SAR442168 formerly known as PRN2246 works by inhibiting Brutons tyrosine. Sanofi SA announced Thursday morning it has inked a deal with Principia Biopharma Inc. French pharma company Sanofi has revealed new long-term data for its investigational oral Brutons tyrosine kinase BTK inhibitor tolebrutinib which demonstrated a promising safety and efficacy profile in patients with relapsing forms of multiple sclerosis MS.
Sanofi Genzyme has launched a website to connect MS patients ages 18-60 with one of its four ongoing clinical trials testing tolebrutinib. Bruton tyrosine kinase BTK is a nonreceptor tyrosine kinase that plays a central role in the signal transduction of the B-cell antigen receptor and other cell surface receptors both in normal and malignant B lymphocytes. June 1 2020 Key Record Dates.
Biogen is betting at least 125 million that it is. Sanofis annual report on Form 20-F for the year ended December 31 2019. The FDA has not yet approved a BTKi MS treatment.
Bruton tyrosine kinase Btk is expressed in B-lymphocytes myeloid cells and platelets and Btk-inhibitors BTKi are used to treat patients with B-cell malignancies developed against autoimmune diseases have been proposed as novel antithrombotic drugs and been tested in patients with severe COVID-19. July 7 2020 Key Record Dates. Sanofi will design the studies to look at the effect of its BTK inhibitor on outcomes including MS relapse rates disability progression and underlying CNS damage.
EFC16035 U1111-1238-1318 Other Identifier. The French pharma will pay 40 million upfront and has agreed to 756 million in milestones as well as royalties. 3 Agenda Introduction Bill Sibold EVP Specialty Care Phase 2 results Erik Wallstroem Therapeutic Area Head Neurology Development Progression in MS and.
EFC16034 U1111-1238-1373 Other Identifier. UTN First Posted. November 2021 Individual Participant Data IPD Sharing Statement.
Is orelabrutinib one of the next big MS therapies. Plan to Share IPD. NCT04458051 Other Study ID Numbers.
The BTK drug may add to Sanofis portfolio though that wouldnt happen for some time. Median time from CLL diagnosis to initiation of venetoclax and BTKi among the 19 patients for whom data were available was 8 years range 3-19 and 11 years range 5-22 respectively. Orelabrutinib is an experimental oral.
Sanofis willingness to bet on a. Median age at BTKi initiation was 72 years range 50-89. November 19 2021 Individual Participant Data IPD Sharing Statement.
Principia Biopharma The Future Worsens Nasdaq Prnb Seeking Alpha
Targeting Bruton Tyrosine Kinase For Multiple Sclerosis Treatment